TMS-Induced Cortical Potentiation during Wakefulness Locally Increases Slow Wave Activity during Sleep
Reto Huber, # Steve K. Esser, # Fabio Ferrarelli, Marcello Massimini, Michael J. Peterson, and Giulio Tononi *
Department of Psychiatry, University of Wisconsin , Madison, Madison , Wisconsin , United States of America
Mick Rugg, Academic Editor
University of California , Irvine , United States of America
# Contributed equally.
* To whom correspondence should be addressed. E-mail: gtononi@wisc.edu
Conceived and designed the experiments: RH GT SE. Performed the experiments: RH SE FF MM MP. Analyzed the data: RH SE. Contributed reagents/materials/analysis tools: RH SE FF MM. Wrote the paper: RH GT.
Received January 11, 2007; Accepted February 13, 2007.
 This article has been cited by other articles in PMC. This article has been cited by other articles in PMC.
Abstract
Background
Sleep slow wave activity (SWA) is thought to reflect sleep need, increasing in proportion to the length of prior wakefulness and decreasing during sleep. However, the process responsible for SWA regulation is not known. We showed recently that SWA increases locally after a learning task involving a circumscribed brain region, suggesting that SWA may reflect plastic changes triggered by learning.
Methodology/Principal Findings
To test this hypothesis directly, we used transcranial magnetic stimulation (TMS) in conjunction with high-density EEG in humans. We show that 5-Hz TMS applied to motor cortex induces a localized potentiation of TMS-evoked cortical EEG responses. We then show that, in the sleep episode following 5-Hz TMS, SWA increases markedly (+39.1±17.4%, p<0.01, n  = =  10). Electrode coregistration with magnetic resonance images localized the increase in SWA to the same premotor site as the maximum TMS-induced potentiation during wakefulness. Moreover, the magnitude of potentiation during wakefulness predicts the local increase in SWA during sleep. 10). Electrode coregistration with magnetic resonance images localized the increase in SWA to the same premotor site as the maximum TMS-induced potentiation during wakefulness. Moreover, the magnitude of potentiation during wakefulness predicts the local increase in SWA during sleep.
Conclusions/Significance
These results provide direct evidence for a link between plastic changes and the local regulation of sleep need. |
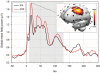
|
Figure 1
Responses to TMS before and after 5 Hz conditioning. Total activation produced by TMS as measured by the global mean field power (GMFP) derived from all 60 electrodes. The GMFP was increased between 10 and 130 ms post stimulus (gray area) following rTMS (more ...) |

|
Table 1
Sleep architecture for the first NREM sleep episode. |

|
Figure 2
Changes in local SWA homeostasis during sleep after 5-Hz rTMS conditioning. A. Topographic distribution of SWA after 5-Hz conditioning (top) and the sham control (bottom) condition. Average EEG power density at 14.5 Hz (n  = =  10 (more ...) 10 (more ...) |
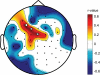
|
Figure 4
The increase in TMS-evoked responses predicts the local increase of SWA. Topographic depiction of positive correlations between SWA change and the change in global mean field power in the late component of the TMS-evoked response after 5-Hz rTMS conditioning. (more ...) |
References
1. Steriade M. The corticothalamic system in sleep. Front Biosci. 2003; 8 :d878d899. [ PubMed ]
2. Steriade M, McCormick DA, Sejnowski TJ. Thalamocortical oscillations in the sleeping and aroused brain. Science. 1993; 262 :679685. [ PubMed ]
3. Borbély, AA.; Achermann, P. Homeostasis of human sleep and models of sleep regulation. In: Kryger MH, Roth T, Dement WC. , editors. Principles and Practice of Sleep Medicine. Philadelphia : W. B. Saunders; 2000. pp. 377390. In:
4.Borbély AA. A two process model of sleep regulation. Human Neurobiol. 1982; 1 :195204.
5. Borbély AA. From slow waves to sleep homeostasis: new perspectives. Arch Ital Biol. 2001; 139 :5361. [ PubMed ] 6.
Huber R, Ghilardi MF, Massimini M, Tononi G. Local sleep and learning. Nature. 2004; 430 :7881. [ PubMed ]
7.
Vyazovskiy V, Borbély AA, Tobler I. Unilateral vibrissae stimulation during waking induces interhemispheric EEG asymmetry during subsequent sleep in the rat. J Sleep Res. 2000; 9 :367371. [ PubMed ]
8.
Huber R, Ghilardi MF, Massimini M, Ferrarelli F, Riedner BA, et al. Arm immobilization causes cortical plastic changes and locally decreases sleep slow wave activity. Nat Neurosci. 2006
9.
Tononi G, Cirelli C. Sleep function and synaptic homeostasis. Sleep Med Rev. 2006; 10 :4962. [ PubMed ]
10.
Hill S, Tononi G. Modeling sleep and wakefulness in the thalamocortical system. J Neurophysiol. 2005; 93 :16711698. [ PubMed ]
11.
Bliss TV, Lomo T. Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. J Physiol. 1973; 232 :331356. [ PubMed ]
12.
Esser SK, Huber R, Massimini M, Peterson MJ, Ferrarelli F, et al. A direct demonstration of cortical LTP in humans: A combined TMS/EEG study. Brain Res Bull. 2006; 69 :8694. [ PubMed ]
13.
Peinemann A, Reimer B, Loer C, Quartarone A, Munchau A, et al. Long-lasting increase in corticospinal excitability after 1800 pulses of subthreshold 5 Hz repetitive TMS to the primary motor cortex. Clin Neurophysiol. 2004; 115 :15191526. [ PubMed ]
14.
Quartarone A, Bagnato S, Rizzo V, Morgante F, Sant'angelo A, et al. Distinct changes in cortical and spinal excitability following high-frequency repetitive TMS to the human motor cortex. Exp Brain Res. 2005; 161 :114124. [ PubMed ]
15.
Finelli LA, Borbély AA, Achermann P. Functional topography of the human nonREM sleep electroencephalogram. Eur J Neurosci. 2001; 13 :22822290. [ PubMed ]
16.
Werth E, Achermann P, Borbély AA. Fronto-occipital EEG power gradients in human sleep. J Sleep Res. 1997; 6 :102112. [ PubMed ]
17.
Frenkel MY, Sawtell NB, Diogo AC, Yoon B, Neve RL, et al. Instructive effect of visual experience in mouse visual cortex. Neuron. 2006; 51 :339349. [ PubMed ]
18.
Malinow R, Mainen ZF, Hayashi Y. LTP mechanisms: from silence to four-lane traffic. Curr Opin Neurobiol. 2000; 10 :352357. [ PubMed ]
19.
Ziemann U. TMS induced plasticity in human cortex. Rev Neurosci. 2004; 15 :253266. [ PubMed ]
20.
Tegenthoff M, Ragert P, Pleger B, Schwenkreis P, Forster AF, et al. Improvement of tactile discrimination performance and enlargement of cortical somatosensory maps after 5 Hz rTMS. PLoS Biol. 2005; 3 :e362. [ PubMed ]
21.
Huang YZ, Edwards MJ, Rounis E, Bhatia KP, Rothwell JC. Theta burst stimulation of the human motor cortex. Neuron. 2005; 45 :201206. [ PubMed ]
22.
Pascual-Leone A, Valls-Sole J, Wassermann EM, Hallett M. Responses to rapid-rate transcranial magnetic stimulation of the human motor cortex. Brain. 1994; 117 ( Pt 4) :847858. [ PubMed ]
23.
Di Lazzaro V, Oliviero A, Berardelli A, Mazzone P, Insola A, et al. Direct demonstration of the effects of repetitive transcranial magnetic stimulation on the excitability of the human motor cortex. Exp Brain Res. 2002; 144 :549553. [ PubMed ]
24.
Kole MH, Fuchs E, Ziemann U, Paulus W, Ebert U. Changes in 5-HT1A and NMDA binding sites by a single rapid transcranial magnetic stimulation procedure in rats. Brain Res. 1999; 826 :309312. [ PubMed ]
25.
Cooke SF, Bliss TV. Plasticity in the human central nervous system. Brain. 2006; 129 :16591673. [ PubMed ]
26.
Ilmoniemi RJ, Virtanen J, Ruohonen J, Karhu J, Aronen HJ, et al. Neuronal responses to magnetic stimulation reveal cortical reactivity and connectivity. Neuroreport. 1997; 8 :35373540. [ PubMed ]
27.
Massimini M, Ferrarelli F, Huber R, Esser SK, Singh H, et al. Breakdown of cortical effective connectivity during sleep. Science. 2005; 309 :22282232. [ PubMed ]
28.
Clapp WC, Zaehle T, Lutz K, Marcar VL, Kirk IJ, et al. Effects of long-term potentiation in the human visual cortex: a functional magnetic resonance imaging study. Neuroreport. 2005; 16 :19771980. [ PubMed ]
29.
Teyler TJ, Hamm JP, Clapp WC, Johnson BW, Corballis MC, et al. Long-term potentiation of human visual evoked responses. Eur J Neurosci. 2005; 21 :20452050. [ PubMed ]
30.
Siebner HR, Peller M, Willoch F, Minoshima S, Boecker H, et al. Lasting cortical activation after repetitive TMS of the motor cortex: a glucose metabolic study. Neurology. 2000; 54 :956963. [ PubMed ]
31.
Loo CK, Mitchell PB. A review of the efficacy of transcranial magnetic stimulation (TMS) treatment for depression, and current and future strategies to optimize efficacy. J Affect Disord. 2005; 88 :255267. [ PubMed ]
32.
Fink GR, Frackowiak RS, Pietrzyk U, Passingham RE. Multiple nonprimary motor areas in the human cortex. J Neurophysiol. 1997; 77 :21642174. [ PubMed ]
33.
Morecraft RJ, Van Hoesen GW. Frontal granular cortex input to the cingulate (M3), supplementary (M2) and primary (M1) motor cortices in the rhesus monkey. J Comp Neurol. 1993; 337 :669689. [ PubMed ]
34.
Tokuno H, Nambu A. Organization of nonprimary motor cortical inputs on pyramidal and nonpyramidal tract neurons of primary motor cortex: An electrophysiological study in the macaque monkey. Cereb Cortex. 2000; 10 :5868. [ PubMed ]
35.
Bestmann S, Baudewig J, Siebner HR, Rothwell JC, Frahm J. Functional MRI of the immediate impact of transcranial magnetic stimulation on cortical and subcortical motor circuits. Eur J Neurosci. 2004; 19 :19501962. [ PubMed ]
36.
Ferrarelli F, Haraldsson HM, Barnhart TE, Roberts AD, Oakes TR, et al. A [17F]-fluoromethane PET/TMS study of effective connectivity. Brain Res Bull. 2004; 64 :103113. [ PubMed ]
37.
Esser SK, Hill SL, Tononi G. Modeling the effects of transcranial magnetic stimulation on cortical circuits. J Neurophysiol. 2005; 94 :622639. [ PubMed ]
38.
Tononi G, Cirelli C. Sleep and synaptic homeostasis: a hypothesis. Brain Res Bull. 2003; 62 :143150. [ PubMed ]
39.
Matsunaga K, Maruyama A, Fujiwara T, Nakanishi R, Tsuji S, et al. Increased corticospinal excitability after 5 Hz rTMS over the human supplementary motor area. J Physiol. 2005; 562 :295306. [ PubMed ]
40.
Ghilardi MF, Ghez C, Dhawan V, Moeller J, Mentis M, et al. Patterns of regional brain activation associated with different forms of motor learning. Brain Res. 2000; 871 :127145. [ PubMed ]
41.
Wassermann EM. Risk and safety of repetitive transcranial magnetic stimulation: report and suggested guidelines from the International Workshop on the Safety of Repetitive Transcranial Magnetic Stimulation, June 57, 1996. Electroencephalogr Clin Neurophysiol. 1998; 108 :116. [ PubMed ]
42.
Lehmann D, Skrandies W. Reference-free identification of components of checkerboard-evoked multichannel potential fields. Electroencephalogr Clin Neurophysiol. 1980; 48 :609621. [ PubMed ]
43.
Rechtschaffen, A.; Kales, A. Bethesda , Maryland : National Institutes of Health; 1968. A manual of standardized terminology, techniques and scoring system for sleep stages of human subjects.
44.
Huber R, Graf T, Cote KA, Wittmann L, Gallmann E, et al. Exposure to pulsed high-frequency electromagnetic field during waking affects human sleep EEG. Neuroreport. 2000; 11 :33213325. [ PubMed ]
45.
Nichols TE, Holmes AP. Nonparametric permutation tests for functional neuroimaging: a primer with examples. Hum Brain Mapp. 2001; 15 :125. [ PubMed ]
46.
Pascual-Marqui RD. Standardized low-resolution brain electromagnetic tomography (sLORETA): technical details. Methods Find Exp Clin Pharmacol. 2002;(24 Suppl D):512. [ PubMed
|
